(c) What-if-Scenario: How would the results change if (c) the compressed liquid table or (d) the solid/liquid model were used to model liquid water instead? Īnswers: (a) 0oC, (b) 5.79 kJ/kg, (c) 5.86 kJ/kg, 5.01 kJ/kgĮXAMPLE E3-14 A piston cylinder device having a volume ofĢ L contains argon at a pressure of 1 atm and a temperature of 550oR. Determine (a) the change in temperature, and (b) enthalpy. (b) Can you explain why entropy remains constant despite an increase in temperature at the exxit? Answers:(a) 48.91oC, (b) 1.02 MPaĮXAMPLE E3-13 Water at 5 MPa and 140oC is pressurized atĬonstant entropy to a pressure of 10 MPa. If the exit pressure is 1 MPa, determine (a) the exit temperature. Into an isentropic (constant entropy) compressor at a pressure of 100 kPa. (c) What-ifScenario: How would the answers change if the tank were heated until the liquid volume reduced to 200 mL? Answers: (a) 3148 kPa, (b) 67.8oC, (c) 2517 kPaĮXAMPLE E3-12 Saturated vapor of refrigerant R-134a flows Determine (a) the pressure, and (b) temperature in the tank. Answers:(a) 35.12%, (b) 678.6 kJ/kg, (c) 540mLĮXAMPLE E3-11 The tank in the last example is heated untilĪll the liquid completely vaporizes. U, and (c) the volume fraction y occupied by the liquid phase. Answers: Saturated MixtureĮXAMPLE E3-10 In the previous problem determine (a) x, (b) Answers: 112.86oCĮXAMPLE E3-9 A rigid tank with a volume of 0.02 m 3 containsĠ.5 kg of refrigerant ammonia (NH3) at 25oC. If the specific enthalpy is 2700 kJ/kg, determine the temperature of the steam. ĮXAMPLE E3-8 A tank contains superheated steam at 120 Assuming the atmospheric conditions to be 100 kPa and 25oC, determine (a) the direction of flow at the leak, and (b) the temperature of the refrigerant when exposed to the atmosphere. What-if-Scenario: How would the answers change if the pressure were increased to 1 MPa? Answers:(a) 99.61oC, (b) -33.63oC, (c) -26.58oC, (d) -195oC, (e) 24.89oCĮXAMPLE E3-7 A pipe carrying refrigerant R-12 at 25oC as a Water, (b) ammonia, (c) R-134a, and (d) nitrogen at 100 kPa. Answers: (a) 0 kJ/kg, (b) 0.11 kJ/kg, (c) 0.90 kJ/kg, (d) 1.01 kJ/kgĮXAMPLE E3-6 Calculate the saturation temperature of (a)

Determine the change in (a) specific internal energy, (b) specific stored energy, (c) specific enthalpy, and (d) specific flow energy between the inlet and exit. At the inlet p1=100 KPa, V1 =10 m/s and z1 =0 and at the exit p2 = 1000 KPa, V2 = 15 m/s and z2 = 5 m. Higher pressure in an isentropic (constant entropy) manner. Determine the change in (a) the internal energy, (b) enthalpy, (c) entropy of the block after itĬomes to equilibrium with the chamber pressure and temperature What-if-scenario: How would the answer in part (a) and (c) change if the block were made of (e) copper, (f) gold? Īnswers: (a) 765 kJ, (b) 783.3 kJ, (c) 2.0 kJ/K, (d) 157.7 kJ, (e) 328.1 kJ, (f) 109.7 kJĮXAMPLE E3-5 A pumping system delivers liquid water at a (b) What-if-scenario: How would the answer change if steam had velocities of 10 m/s and 100 m/s in the two states? Answers: (a) 5.3%, (b) 5.3%ĮXAMPLE E3-4 A block of aluminum of mass 5 kg, initially atĮquilibrium with the surroundings at 30oC and 100 kPa, is placed in a pressurized chamber kept at a pressure of 10 MPa and a temperature of 200 oC.

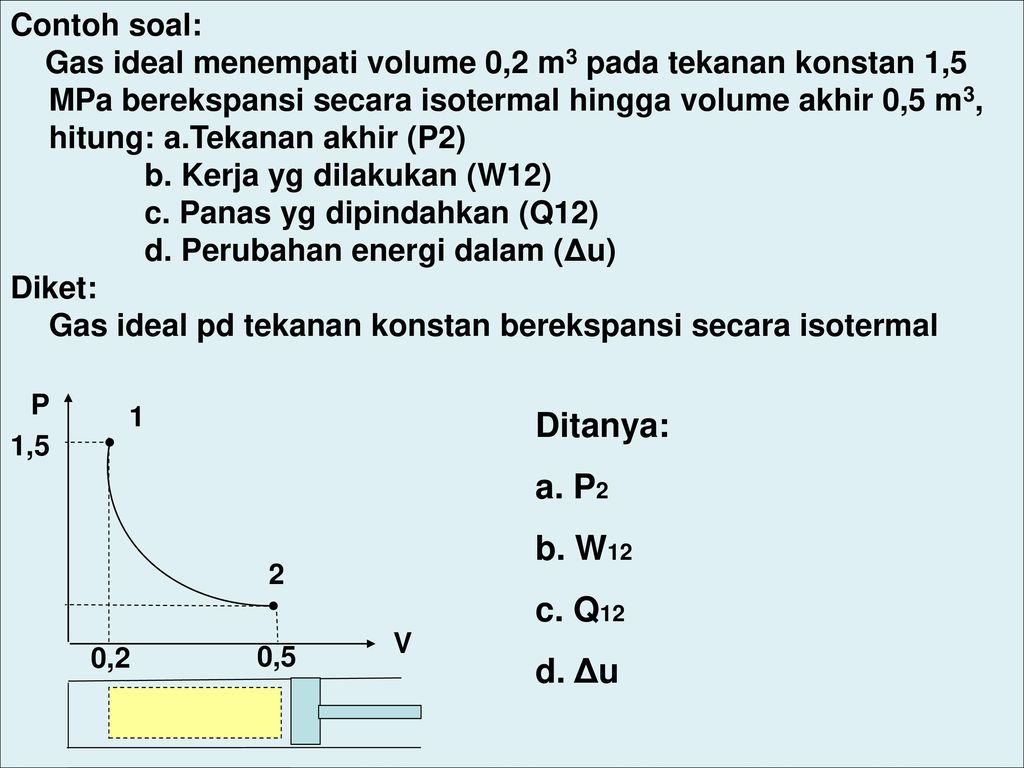
#CONTOH SOAL GAS IDEAL PC#
Use PC (Phase-Change) system state daemon. Evaluate (a) the difference between the left and right hand sides of the first T-ds equation if the system comes to a new equilibrium with its pressure decreasing by 1 kPa and temperature increasing by 1oC. Pressure of 1 MPa and a temperature of 300oC. Answers: (a) 0.619m3/kg, (b) 0.615m3/kgĮXAMPLE E3-3 A piston-cylinder device contains steam at a Determine the change in, (a) exactly and (b) approximately using Taylor's theorem for the following change of state. Answers: (a) 0.895 m3/kg, 3.11 kJ/kg, (b) 0.02 kWĮXAMPLE E3-2 The specific volume of a gas in m3/kg isĮxperimentally related to the independent properties temperature and pressure through the following functional relationship: = 4.157 T/p, where T and p are expressed in K and kPa respectively. If the nozzle exit diameter is 1 cm, determine (a) two thermodynamic properties at the exit, and (b) the flow rate of enthalpy. Soal soal dibawah ini merupakan merupakan contoh penerapan dari konsep yang teah dijelaskan dalam pertemuan ke 4 dan 5, baik substansi materi yang tertuang dalam e learning termo maupun saat perkuliahan tatap muka di ruang kelasEXAMPLE E3-1 At the exit of a nozzle (see accompanyingįigure) a gas has the following properties: V1 = 95 m/s, mdot2 = 0.5 kg/min, Jdot2 = 0.0635 kW.
